LIPSTATIN/ORLISTAT
PopularDescription
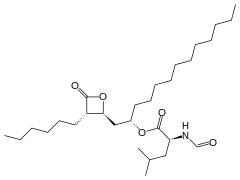
Member of Farmavita.net offers know-how for manufacture of active pharmaceutical ingredient of orlistat.
Orlistat (marketed under the trade name Xenical by Roche; or over-the-counter as alli by GlaxoSmithKline (pronounced /ˈælaɪ/, like the English word "ally")-also known as tetrahydrolipstatin-is a drug designed to treat obesity. Its primary function is preventing the absorption of fats from the human diet, thereby reducing caloric intake. It is intended for use in conjunction with a physician-supervised reduced-calorie diet.
Orlistat is the saturated derivative of lipstatin-a potent natural inhibitor of pancreatic lipases isolated from the bacterium Streptomyces toxytricini. However, due to simplicity and stability, orlistat rather than lipstatin was developed into an anti-obesity drug.
Know how for manufacture is available as non-exclusive license with lump sum payment. Payment is made after yield and quality of API is confirmed in the laboratory. Guarantied process parameters are:
Final titre 7+ 1 g/l
Fermentation time 192 hrs
Recovery yield: > 70%
Offer is valid subject to confirmation, country by country. There will be no sales in those countries where this offer would constitute an infringement of third parties intellectual proprietary rights. Farmavita.Net is not owner of offered products/licenses and do not take any liability related to them. However, as the licensing agent we do our the best to facilitate that closed licensing agreements are made on mutual benefit of contracting parties and without infringement of intellectual property of third parties.
More details
| Name | Description |
|---|---|
| Pharma licensing | Out-licensing (offer) |

